Oncogenic lncRNAs as markers of breast cancer progression
The p53 family member and p63 isoform TAp63 is a crucial tumor and metastasis suppressor whose loss leads to highly metastatic breast adenocarcinomas in mice and correlate with high-grade breast cancers in humans. By performing a mouse-human cross species analysis, we identified a group of oncogenic long non-coding RNAs (lncRNAs), whose expression is controlled by TAp63 and correlates with breast cancer evolution and tumor grade. By analyzing the common mechanism of function of these lncRNAs, we have obtained molecular, functional, and translational evidence of the tumorigenic and metastatic potential of these lncRNAs and their suitability as prognostic markers of breast cancer progression.
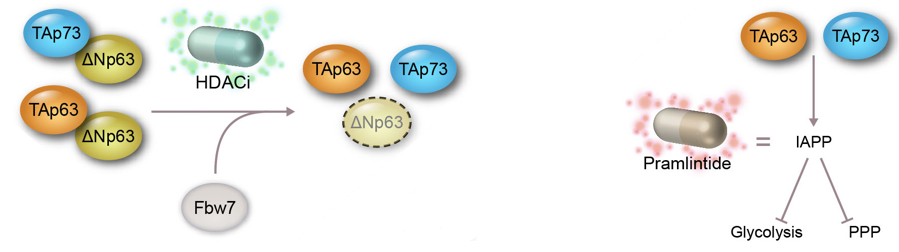
Targeting the p53 pathway therapeutically
The tumor suppressor TP53 is mutated in up to 50% of human cancers, while the remainder of these cancers exhibit inactivation of the p53 pathway via alternative mechanisms. All the proposed therapies to re-activate p53 functions have failed so far. Therefore, we evaluated the efficacy of targeting the other p53 family members, p63 and p73. Through this approach, we identified 2 mechanisms to counteract the pro-oncogenic activities of ∆Np63 and ∆Np73 in tumors addicted to such factors, thus enhancing the tumor-suppressive activities of TAp63 and TAp73: i) HDAC inhibitors reducing the protein stability of ∆Np63; and ii) pramlintide, a FDA approved therapy for diabetes, successfully opposing the metabolic reprogramming induced by ∆Np63 and ∆Np73.
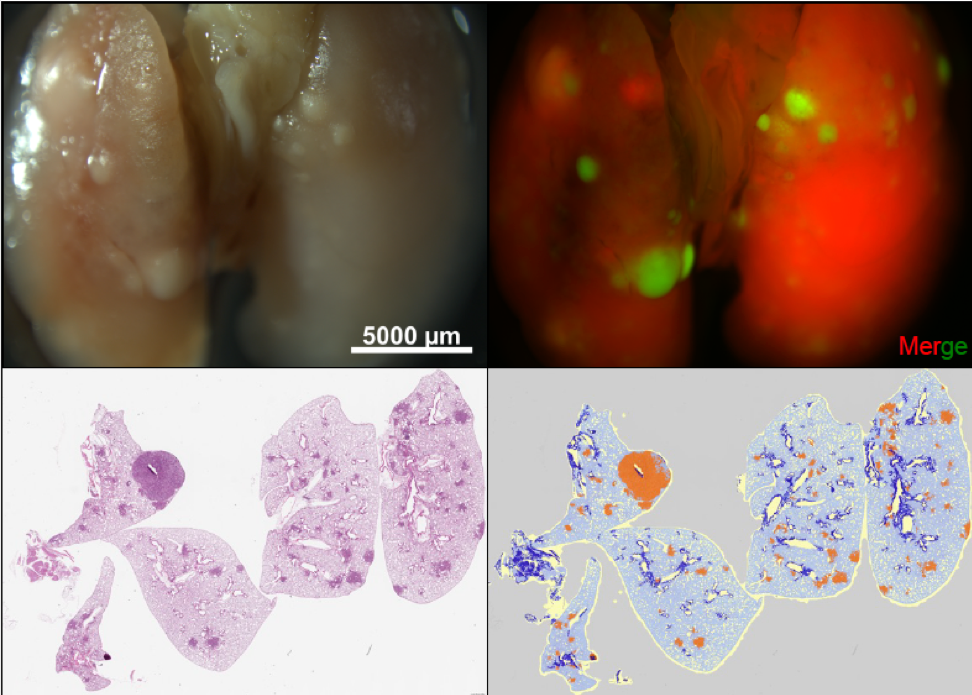
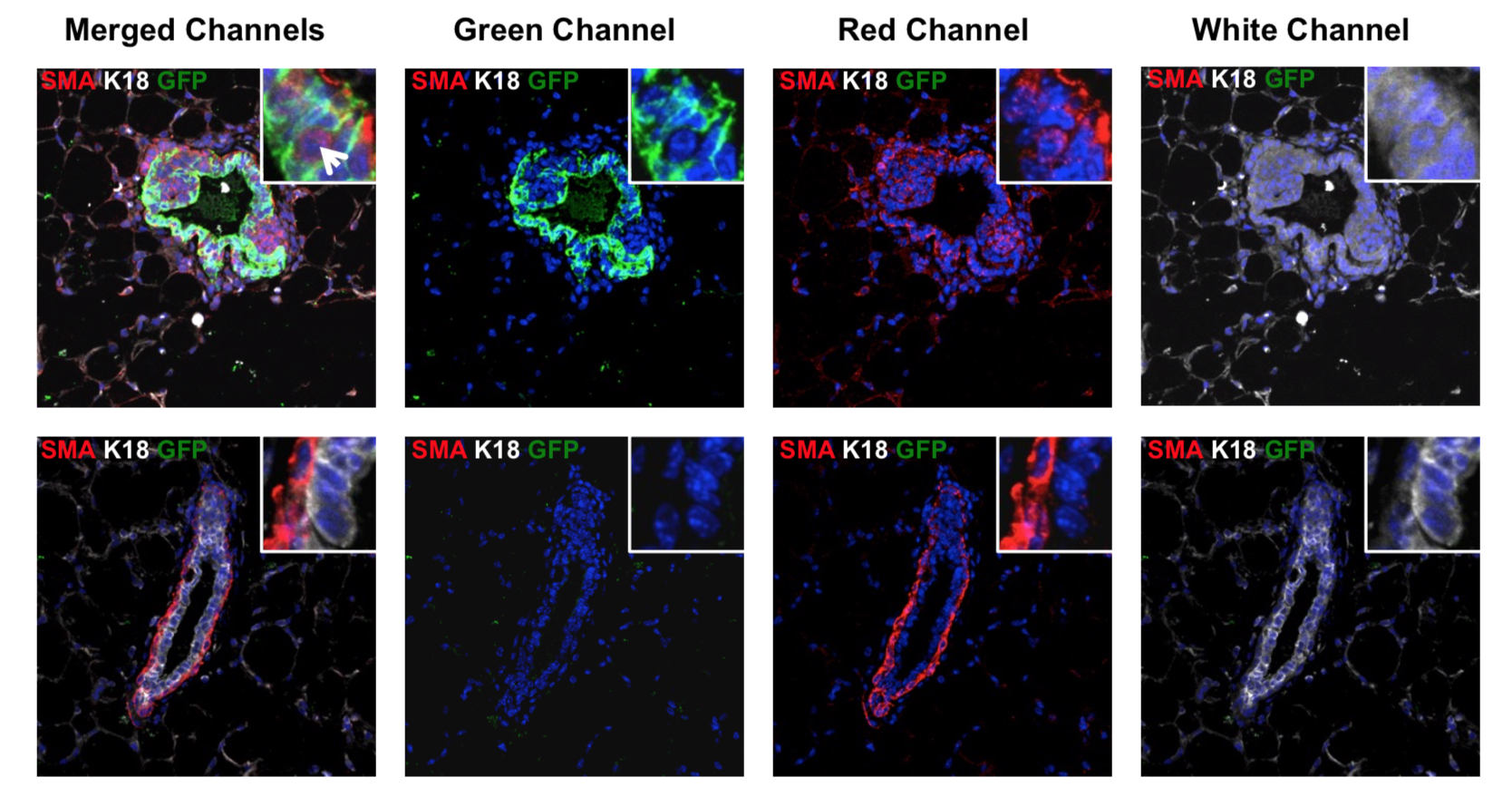
Role of TAp73 in lung adenocarcinoma
Our laboratory is working to elucidate the roles of the p53 family of transcription factors in cancer development, progression, and metastasis of various types of tumors. Mutant p53 has been shown to extensively interact with p73 and its tumor suppressive isoform TAp73 to inhibit its tumor suppressive functions. To assess the role of TAp73 in lung tumorigenesis and progression, our laboratory developed a TAp73 conditional knockout mouse where exons 2 and 3 of TAp73 are flanked by loxP sites (TAp73fltd/fltd). These mice were intercrossed to KrasLSLG12D/+ mice carrying a mutation on the Kras gene making the protein constitutively active upon Cre recombination. The intratracheal delivery of adenovirus expressing Cre recombinase into these mice results in the formation of multi-focal aggressive lung adenocarcinomas. Interestingly, we found that deletion of TAp73 in the context of oncogenic Kras promoted tumor development and progression. Ongoing studies in the laboratory seek to investigate the mechanisms employed by TAp73 in tumor initiation and progression of lung adenocarcinoma. Projects also include investigating the function of p73 in small cell lung cancer. We are currently investigating functions of p73 in tumor metabolism, regulation of non-coding RNAs and tumor immunity.